News
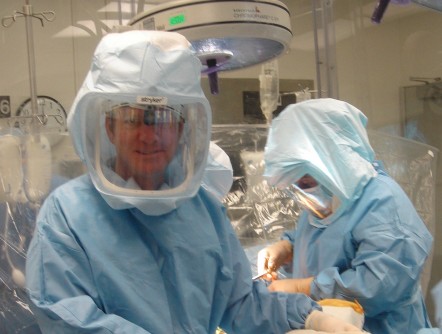
Newswise — In a career spanning more than two decades, orthopedic surgeon Geoffrey Westrich has seen numerous advances in joint replacement surgery that benefit patients. He believes robotic-assisted knee replacement, which allows for an ultraprecise procedure, is one of the most exciting.
Dr. Westrich, who specializes in knee and hip replacement at Hospital for Special Surgery in New York City, reached a milestone last year when he performed his 500th robotic-assisted knee replacement. He uses the MAKO robotic system, which is FDA-approved for both total and partial knee replacements.
"Joint replacement is highly successful in relieving arthritis pain and improving quality of life, and the robotic system allows us to really customize the procedure for each patient," says Dr. Westrich. “The MAKO system allows for optimal alignment and positioning of the knee implant, as well as optimal ligament balancing, all critically important for the best outcome and long-term success of the surgery. Such precision could potentially lead to a longer-lasting knee replacement.”
Dr. Westrich says over the past few years, he has seen more patients in their 40s and 50s with arthritis who are candidates for a knee replacement. The main concern for patients in this age group is that the implant may wear out over time. Although a knee replacement could last 20 years, or even longer, it doesn’t last indefinitely. For younger individuals in particular, robotic-assisted surgery has made joint replacement an attractive option, as it could prolong the life of the implant, according to Dr. Westrich.
Over the past couple of years, studies have focused on the pinpoint accuracy of the robotic-assisted knee replacement system. A study by Dr. Westrich and colleagues published in the Journal of Knee Surgery this year found that the technology was advantageous in the training of orthopedic surgeons, as it allowed for increased accuracy and precision.
A separate study in the Bone & Joint Journal in 2018 by researchers in the United Kingdom found robotic-assisted total knee replacement was associated with decreased pain after surgery, improved early functional recovery and a shorter hospital stay.
Before surgery, a CT scan is taken of the patient’s knee. The scan is then uploaded into the Mako system software, where a 3D model of the joint is created. The 3D model is used to plan and assist the surgeon in performing the joint replacement.
In the operating room, the orthopedic surgeon controls a robotic arm that uses computer‐guided mapping software, similar to GPS, integrated into the surgical instruments to position the implant in the knee joint.
The digital tracking system constantly monitors and updates the patient’s anatomy and enables the surgeon to make real‐time adjustments to optimize implant placement, alignment, ligament balance and joint motion. This provides each patient with a personalized surgery tailored to his or her individual anatomy.
“With more accurate alignment and positioning, the implant should experience less wear and friction, and it could ultimately last longer,” says Dr. Westrich, who notes that studies will be needed to confirm this over the long term.
He says the precision of the robotic‐assisted procedure also offers more protection to the surrounding soft tissues and enables more healthy bone to be preserved, an advantage in the event another knee replacement is needed down the road.
Newswise — The overuse of antibiotics occurs due to the mistaken widespread belief that they are beneficial for a broad array of conditions and because many physicians are willing to prescribe antibiotics if patients ask for the medication, according to a Rutgers study.
The study, published in the journal BioEssays, reviewed more than 200 peer-reviewed studies to examine the causes behind antibiotic overuse, which can lead harmful bacteria to become drug-resistant and cause harmful effects on the microbiome, the collection of beneficial germs that live in and on our bodies.
Martin Blaser, director of the Center for Advanced Biotechnology and Medicine at Rutgers and lead author, said the global use of antibiotics between 2000 and 2015 increased 39 percent, with a 77 percent increase in low- and middle-income countries. He discusses the study’s findings.
What health concerns result from the disruption of the microbiome by antibiotics?In children, improper antibiotic use can alter the microbiome while their immunological, metabolic and neural systems are developing. Epidemiological studies associate antibiotic exposure with an increased risk of disease of allergic, metabolic and cognitive disorders that have grown more common in children during the antibiotic era.
In adults, there is increasing evidence that antibiotics may enhance risk for metabolic and neoplastic diseases, including diabetes, kidney stones and growths in the colon and rectum that can lead to cancer.
What are the trends you found in antibiotic use?Studies in the United States, United Kingdom and China found numerous online pharmacies selling antibiotics without a prescription. This problem also is large in Iowa- to middle-income countries, where 60 percent of antibiotics are sold without prescriptions, often by untrained medical practitioners.
Perhaps of special concern during the COVID-19 pandemic is the finding that telemedicine services are another potential source of questionable antibiotic sales in the United States. A recent analysis found that patients with acute respiratory infections were more often prescribed broad-spectrum antibiotics if they had a tele-health doctor visit, compared to an in-person visit.
Worldwide, antibiotic use is highest in young children, especially in low-income areas. This is often in response to the fact that young children are prone to have four to six upper respiratory tract infections each year. Although most of these infections are treated by antibiotics, 80 percent are not caused by bacteria and would therefore derive no benefit from antibiotics.
Are some practitioners more likely to prescribe antibiotics?Our findings are consistent with the hypothesis that older physicians are more likely than their younger colleagues to prescribe antibiotics. For example, one study found that physicians over 30 were several times more likely to prescribe antibiotics for common respiratory conditions that do not necessarily require them. Another study found that physicians with over 25 years in practice were disproportionately likely to issue prescriptions of more than eight days.
What misinformation did you find among the public?Many people believe that antibiotics are effective against bacterial and viral illnesses, lumping all types of pathogens together and adopting a “germs are germs” attitude. Others believe that taking antibiotics can’t hurt. Across Europe, for example, 57 percent of people surveyed were unaware that antibiotics were ineffective against viruses, and 44 percent did not know that antibiotics have no effect against colds or influenza.
What other reasons did you find for inappropriate prescription of antibiotics?Antibiotics are commonly used across the world to self-treat health problems for which they were never intended, such as in Nigeria, where women are increasingly using antibiotics to reduce menstrual cramps. In low- to middle-income countries, antibiotics are often seen as strong, magical medicines, capable of both curing and preventing a range of illness. In many countries people also take them to return to work or school when ill. One of the studies found that 63 percent of Chinese university students kept a personal antibiotic stock at home.
Parents may appeal for an antibiotic for their children so that they can go to work or for the children to return to school or daycare. A U.S. study found that 43 percent of parents of a child with cold symptoms believed that antibiotics were necessary.
In addition, some doctors are inclined to prescribe an antibiotic to maintain a good relationship with patients who expect to receive medication. Patients may not demand antibiotics outright, but rather infer their need for them by how they describe the severity of their illness or note that they worked in the past for a similar issue. People have become less willing to wait and let an illness run its course. The perception that there is a pill for ills of all kinds leads the public to demand immediate relief for symptoms from practitioners and to self-medicate.
Every time an antibiotic is given, money changes hands. This is especially a problem in low- and middle-income countries, where pharmacists are happy to dispense without a prescription to their customers. The rural health practitioners in China are paid every time they dispense an antibiotic as well. Such monetary incentives favor the wide use of antibiotics.
How can antibiotic overuse be addressed?Clinicians need to be better educated about the long-term effects on the microbiome and learn about better ways to speak with their patients about antibiotic risks and benefits. They also need to improve their communication about the consequences of antibiotic treatments and identify antibiotic alternatives.
Blaser conducted the study with three eminent anthropologists (Melissa Melby, University of Delaware; Margaret Lock, McGill University; and Mark Nichter, University of Arizona). All are members of the Canadian Institute for Advanced Research (CIFAR).
One form of immunotherapy weakens cancer and renders it vulnerable to another form, creating one-two punch
Newswise — HAMILTON, ON, Jan. 21, 2020 — McMaster University researchers have established in lab settings that a novel combination of two forms of immunotherapy can be highly effective for treating lung cancer, which causes more deaths than any other form of cancer.
The new treatment, yet to be tested on patients, uses one form of therapy to kill a significant number of lung tumor cells, while triggering changes to the tumor that enable the second therapy to finish the job.
The first therapy employs suppressed “natural killer” immune cells by extracting them from patients’ tumours or blood and supercharging them for three weeks. The researchers condition the cells by expanding and activating them using tumour-like feeder cells to improve their effectiveness before sending them back into battle against notoriously challenging lung tumors.
The supercharged cells are very effective on their own, but in combination with another form of treatment called checkpoint blockade therapy, create a potentially revolutionary treatment.
“We’ve found that re-arming lung cancer patients’ natural killer immune cells acts as a triple threat against lung cancer,” explains Sophie Poznanski, the McMaster PhD student and CIHR Vanier Scholar who is lead author of a paper published today in the Journal for ImmunoTherapy of Cancer.
“First, these highly activated cells are able to kill tumour cells efficiently. Second, in doing so, they also reactivate tumour killing by exhausted immune cells within the patients’ tumours. And third, they release factors that sensitize patients’ tumours to another immunotherapy called immune checkpoint blockade therapy.
“As a result, we’ve found that the combination of these two therapies induces robust tumour destruction against patient tumours that are initially non-responsive to therapy.”
Previous breakthroughs in checkpoint blockade therapy had earned Japanese researcher Tasuku Honjo and American immunologist James Allison the 2018 Nobel Prize for Medicine or Physiology.
Checkpoint blockade therapy works by unlocking cancer’s defence against the body’s natural immune response. The therapy can be highly effective in resolving even advanced cases of lung cancer – but it only works in about 10 per cent of patients who receive it.
The research team, featuring 10 authors in total, has shown that the supercharged immune cells, when deployed, release an agent that breaks down tumors’ resistance to checkpoint blockade therapy, allowing it to work on the vast majority of lung-cancer patients whose tumors would otherwise resist the treatment.
Once activated, the natural killer cells are able to secrete inflammatory factors that help enhance the target of the blockchain that the other immunotherapy treats.
“We needed to find a one-two punch to dismantle the hostile lung tumor environment,” says Ali Ashkar, a professor of Medicine and a Canada Research Chair who is Poznanski’s research supervisor and the corresponding author on the paper. “Not only is this providing a new treatment for hard-to-treat lung cancer tumors with the natural killer cells, but that treatment also converts the patients who are not responsive to PD1-blockade therapy into highly responsive candidates for this effective treatment”.
Such progress is possible because of the close collaboration among clinical practitioners and lab-based researchers at McMaster and its partner institutions, Ashkar says.
He said the team’s clinical practitioners, who work with cancer patients every day, provided critical wisdom and collected vital samples from patients at St. Joseph’s Healthcare Hamilton. Ashkar says those clinicians’ insights and the samples were integral to the research.
Co-author Yaron Shargall, a professor in and Chief of the Division of Thoracic Surgery at McMaster’s Michael G. DeGroote School of Medicine and a thoracic surgeon at St. Joseph’s Healthcare Hamilton, says the promising outcome is the result of close links between basic science and clinical medicine.
“It was successful mostly due to the facts that the two groups have spent long hours together, discussing potential ways of combining forces and defining a linkage between a highly specific basic science technology and a very practical clinical, day-to-day dilemmas,” he said. “This led to a flawless collaboration which resulted in a very elegant, potentially practice-changing, study.”
The researchers are now working to organize a human clinical trial of the combined therapies, a process that could be under way within months, since both immunotherapies have already been approved for individual use.
Photo info:
Georgia Kirkos, McMaster University
McMaster University researchers Ali Ashkar and Sophie Poznanski.
Newswise — One of the most vexing aspects of the COVID-19 pandemic is doctors’ inability to predict which newly hospitalized patients will go on to develop severe disease, including complications that require the insertion of a breathing tube, kidney dialysis or other intensive care. Knowledge of a patient’s age and underlying medical conditions can help predict such outcomes, but there are still surprises when younger, seemingly healthier patients suffer severe complications that can lead to death.
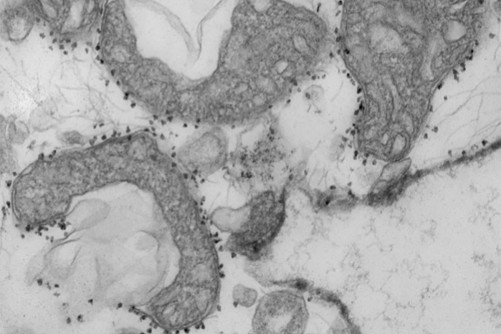
Now, scientists at Washington University School of Medicine in St. Louis have shown that a relatively simple and rapid blood test can predict — within a day of a hospital admission — which patients with COVID-19 are at highest risk of severe complications or death.
The study, published Jan. 14 in JCI Insight, involved nearly 100 patients newly admitted to the hospital with COVID-19.
The blood test measures levels of mitochondrial DNA, a unique type of DNA molecule that normally resides inside the energy factories of cells. Mitochondrial DNA spilling out of cells and into the bloodstream is a sign that a particular type of violent cell death is taking place in the body.
“Doctors need better tools to evaluate the status of COVID-19 patients as early as possible because many of the treatments — such as monoclonal antibodies — are in short supply, and we know that some patients will get better without intensive treatments,” said co-senior author Andrew E. Gelman, PhD, the Jacqueline G. and William E. Maritz Endowed Chair in Immunology and Oncology in the Department of Surgery.
“There’s so much we still don’t understand about this disease,” he added. “In particular, we need to understand why some patients, irrespective of their ages or underlying health in some cases, go into this hyperinflammatory death spiral. Our study suggests that tissue damage may be one cause of this spiral, since the mitochondrial DNA that is released is itself an inflammatory molecule.”
The researchers said the test could serve as a way to predict disease severity as well as a tool to better design clinical trials, identifying patients who might, for example, benefit from specific investigational treatments. They also said they would like to evaluate whether the test could serve as a way to monitor the effectiveness of new therapies. Presumably, effective treatments would lower mitochondrial DNA levels.
“We will need larger trials to verify what we found in this study, but if we could determine in the first 24 hours of admission whether a patient is likely to need dialysis or intubation or medication to keep their blood pressure from dropping too low, that would change how we triage the patient, and it might change how we manage them much earlier in the disease course,” said co-senior author Hrishikesh S. Kulkarni, MD, an assistant professor of medicine.
The researchers, including co-first authors Davide Scozzi, MD, PhD, a staff scientist, and Marlene Cano, PhD, a postdoctoral research scholar, evaluated 97 patients with COVID-19 at Barnes-Jewish Hospital, measuring their mitochondrial DNA levels on the first day of their hospital stays. They found that mitochondrial DNA levels were much higher in patients who eventually were admitted to the ICU, intubated or died. The researchers found this association held independently of a patient’s age, sex and underlying health conditions.
On average, mitochondrial DNA levels were about tenfold higher in patients with COVID-19 who developed severe lung dysfunction or eventually died. Those with elevated levels were almost six times more likely to be intubated, three times more likely to be admitted to the ICU and almost twice as likely to die compared with those with lower levels.
Further, the test predicted outcomes as well as or better than existing markers of inflammation currently measured in patients hospitalized with COVID-19. Most other markers of inflammation measured in patients with COVID-19, including those still under investigation, are general markers of systemic inflammation, rather than inflammation specific to cell death, according to the researchers.
“Viruses can cause a type of tissue damage called necrosis that is a violent, inflammatory response to the infection,” Gelman said. “The cell breaks open, releasing the contents, including mitochondrial DNA, which itself drives inflammation. In COVID-19 patients, there has been anecdotal evidence of this type of cell and tissue damage in the lung, heart and kidney. We think it’s possible that measures of mitochondrial DNA in the blood may be an early sign of this type of cell death in vital organs.”
The researchers also emphasized that the test is quick and straightforward to perform in most hospital settings because it uses the same machinery that processes the standard PCR test for COVID-19. The method they developed allows mitochondrial DNA levels to be quantified directly in the blood. Without requiring intermediate steps to extract the DNA from the blood, the technique returned results in less than an hour.
Before they can apply for approval from the Food and Drug Administration (FDA), the scientists will need to verify that the test is accurate in a larger multi-center trial. They have plans to expand the research to more sites.
The study utilized samples obtained from the School of Medicine’s COVID-19 biorepository, which was developed by co-authors Jane O’Halloran, MD, PhD, an assistant professor of medicine; Charles Goss, PhD, an instructor in biostatistics; and Phillip Mudd, MD, PhD, an assistant professor of emergency medicine.
Photo Info:
WANDY BEATTY
A new study from Washington University School of Medicine in St. Louis suggests that measuring mitochondrial DNA in the blood of patients with COVID-19 can help predict which patients are at highest risk of severe disease, requiring more intensive care. Mitochondrial DNA levels are a measure of tissue damage. Pictured are damaged mitochondria (dark grey areas) released from human lungs. The small dark dots surrounding the mitochondria are magnetic beads that carry antibodies used to isolate and study unhealthy mitochondria that have been released from dying tissues.
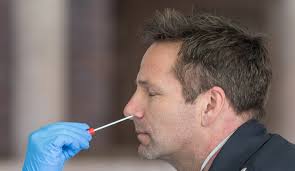
Newswise — HOUSTON-(Jan. 14, 2020) – A new Houston Methodist study out this week found males are more likely to test positive for COVID-19, have complications and die from the virus than females, independent of age.
“Males seem to be more likely to contract the SARS-CoV-2 virus and also have a poor clinical course and outcomes related to COVID-19, compared to females,” said the study’s corresponding author Farhaan Vahidy, Ph.D., M.B.B.S., M.P.H., of the Houston Methodist Research Institute and associate director of the Center of Outcomes Research at Houston Methodist. “The exact contribution of gender and sex factors in susceptibility and outcomes of COVID-19 need further investigation.”
The peer-reviewed observational study appears in PLOS ONE, a multidisciplinary journal published by the Public Library of Science, which is a nonprofit open-access publisher and advocacy organization dedicated to accelerating progress in science and medicine.
As the COVID-19 pandemic unfolds and evolves across the globe, researchers have identified population sub-groups with higher levels of disease vulnerability, such as those with advanced age or certain pre-existing conditions. Small studies from China and Europe have indicated that males tend to experience higher disease severity compared to females. However, a comprehensive gender analysis of COVID-19 in a large and diverse U.S. metropolitan area has been lacking.
To determine the associations between sex and COVID-19 epidemiology, researchers used data from the Houston Methodist COVID-19 Surveillance and Outcomes Registry, which is an IRB-approved observational research registry for COVID-19 patients that tracks socio-demographic, comorbidity, clinical and outcomes data on all individuals tested for SARS-CoV-2 across Houston Methodist’s system of eight hospitals. Data on COVID testing, hospital stays, mortality and demographics were extracted from Electronic Medical Records of all 96,496 adults over 18 who were tested for SARS-CoV-2 by the health system between March 6 and Aug. 22, 2020.
Overall, 15.5% of individuals in the cohort tested positive for SARS-CoV-2. After adjusting for sociodemographic factors and comorbidities, males had a higher likelihood of SARS-CoV-2 positivity than females. Similarly, the proportion of patients requiring ICU care was significantly higher among males at 34.1% as compared to females at 27.6%. Moreover, 19.0% of males underwent mechanical ventilation, compared to 14.7% of females, and the proportion of males who experienced in-hospital mortality was significantly higher at 11.6% as compared to 8.3% of females. The authors conclude that there is a clear and strong independent association between male sex and SARS-CoV-2 susceptibility, complications and poor outcomes and say that understanding gender differences in the disease is a fundamental step toward improved disease management and intervention strategies for both men and women.
The findings are described in a paper titled “Sex differences in susceptibility, severity, and outcomes of coronavirus disease 2019: Cross-sectional analysis from a diverse US metropolitan area,” appearing Jan. 13 in PLOS ONE. Other collaborators working with Vahidy on this study were Alan P. Pan, Hilda Ahnstedt, Yashasvee Munshi, Huimahn A. Choi, Yordanos Tiruneh, Khurram Nasir, Bita A. Kash, Julia D. Andrieni and Louise D. McCullough.
UT Southwestern Medical Center
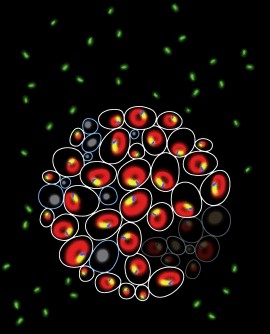
Above are fluorescent images of genetically identical yeast cells, marked for some of the biomarkers that UT Southwestern researchers discovered help to predict cell fate. The yellow is the nucleus, the blue and green are nucleus-vacuole junction (NVJ) markers, and the red is the yeast vacuole.
Biomarkers provide clues to the paths cells take after starvation in findings that may lead to improved drug treatments
Newswise — DALLAS – Jan. 11, 2021 – A set of biomarkers not traditionally associated with cell fate can accurately predict how genetically identical cells behave differently under stress, according to a UT Southwestern study. The findings, published by Cell Reports as a Dec. 1 cover story, could eventually lead to more predictable responses to pharmaceutical treatments.
Groups of the same types of cells exposed to the same stimuli often display different responses. Some of these responses have been linked to slight differences in genetics between individual cells. However, even genetically identical cells can diverge in behavior.
One example can be found in budding yeast, or yeast that are actively dividing. When these microorganisms are deprived of glucose – the sugar molecules they use for energy – all cells stop dividing. However, when this nutrient becomes available again, some cells start dividing once more while others no longer divide but remain alive, even in batches of yeast that are genetic clones. What drives the differences in behavior between these re-dividing “quiescent” cells and never-dividing “senescent” cells has been a mystery, say study leaders N. Ezgi Wood, Ph.D., a postdoctoral fellow at UTSW, and Mike Henne, Ph.D., assistant professor of cell biology and biophysics at UTSW.
UT Southwestern Medical Center
Mike Henne, Ph.D.
Previous studies of behavioral differences in genetically identical cells have focused on genes that decide cell fate. However, Wood, Henne, and their colleagues took a different tact: They looked at the behavior of other biomarkers associated with basic cell maintenance, such as cell cycling, stress response, intracellular communication, and nutrient signaling.
The researchers note that the role each of these factors plays in deciding cell fate is not yet clear. Learning more about the factors that prompt cells to act differently could eventually steer researchers in new directions. For example, the knowledge could be useful in helping cells uniformly respond to cancer chemotherapies or antibiotics, areas in which cells often take divergent paths.
“How two identical cells side by side take different paths is a very basic biological question – we see it from bacteria to mammalian cells,” Wood says. “Our results show that factors not traditionally associated with cell fate can, in fact, play an important role in this process, and gets us closer to answering the question of why this phenomenon takes place and how we might control it.”
To explore these questions, researchers genetically modified yeast cells so that five different protein markers associated with these maintenance tasks glowed with different colors inside the cell when they were present. They then set up an experiment in which these cells lived in a microfluidics chamber that was continuously flushed with liquid media. For two hours, this media was rich with the nutrients that these cells needed to survive and multiply, including glucose. Then, for the next 10 hours, the researchers cut off the glucose supply, starving the cells. At the end of this period, they reintroduced glucose, allowing the cells to recover. During this 16-hour cycle, a camera continuously monitored individual cells, looking for differences between those that became quiescent or senescent when glucose was available again.
When they reviewed the camera footage, researchers quickly saw that despite the cells growing in an asynchronized fashion, or at different points in their cell cycles, starvation stopped the cell cycle. A closer look showed that a protein inhibitor of the cell cycle known as Whi5 tended to collect in the nuclei of quiescent cells during starvation, while Whi5 in senescent cells disappeared altogether.
Similarly, the two populations exhibited differences in the proteins Msn2 and Rtg1 that are associated with stress response. Although these proteins collected in the nuclei of all the cells when they were starved, they had a sustained presence in the nuclei of senescent cells even after glucose returned, yet largely exited the nuclei of quiescent cells when starvation ended.
The researchers found another useful marker for separating these two populations in the nucleus-vacuole junction (NVJ), an interface that connects the nucleus to the vacuole, the small digestive organelle that cells use to sequester waste products. While quiescent cells tended to enlarge their NVJs during starvation, senescent cells did not.
Although each of these findings gave clues to which path cells would take after starvation started, none showed any predictive powers before starvation took place. But when the researchers examined Rim15, a protein that plays a key role in nutrient signaling, they found that cells with elevated Rim15 before starvation tended to become quiescent while those with lower concentrations of this protein were more likely to become senescent.
On their own, none of these factors served as an accurate predictor of cell fate. But when Wood, Henne, and their colleagues performed a statistical analysis incorporating all of them, they were able to accurately predict which cells became quiescent and which became senescent with an accuracy of nearly 90 percent before they reintroduced glucose. In fact, they say, cells seem to reach a “decision point” where it’s unlikely that they’ll change their direction about four hours into starvation.
Other UTSW researchers who contributed to this study include Piya Kositangool, Hanaa Hariri, and Ashley J. Marchand. Henne is a W.W. Caruth, Jr. Scholar in Biomedical Research and a member of the Harold C. Simmons Comprehensive Cancer Center.
This research was funded by grants from the Cancer Prevention and Research Institute of Texas (RR150058), The Welch Foundation (I-1873), National Institutes of Health NIGMS (GM119768), the Ara Parseghian Fund (APMRF2020), and UTSW Endowed Scholars Program.
About UT Southwestern Medical Center
UT Southwestern, one of the premier academic medical centers in the nation, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty has received six Nobel Prizes, and includes 23 members of the National Academy of Sciences, 17 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 2,500 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide care in about 80 specialties to more than 105,000 hospitalized patients, nearly 370,000 emergency room cases, and oversee approximately 3 million outpatient visits a year.
Newswise — In a study to examine a Mediterranean diet in relation to prostate cancer progression in men on active surveillance, researchers from The University of Texas MD Anderson Cancer Center found that men with localized prostate cancer who reported a baseline dietary pattern that more closely follows the key principles of a Mediterranean-style diet fared better over the course of their disease.
“Men with prostate cancer are motivated to find a way to impact the advancement of their disease and improve their quality of life,” said Justin Gregg, M.D., assistant professor of Urology and lead author of the study, published today in Cancer. “A Mediterranean diet is non-invasive, good for overall health and, as shown by this study, has the potential to effect the progression of their cancer.”
After adjusting for factors known to increase risk of cancer getting worse over time, such as age, prostate-specific antigen (PSA) and tumor volume, men with a diet that contained more fruits, vegetables, legumes, cereals and fish had a reduced risk of their prostate cancer growing or advancing to a point where many would consider active treatment. The researchers also examined the effect of diabetes and statin use and found a similar risk reduction in these patient groups.
The study, whose largest number of participants were white, also found that the effect of a Mediterranean diet was more pronounced in African American participants and others who self-identified as non-white. These findings are significant as the rate of prostate cancer diagnosis is more than 50% higher in African American men, who also have a higher risk of prostate cancer death and disease progression.
“The Mediterranean diet consistently has been linked to lower risk of cancer, cardiovascular disease and mortality. This study in men with early stage prostate cancer gets us another step closer to providing evidence-based dietary recommendations to optimize outcomes in cancer patients, who along with their families, have many questions in this area,” said Carrie Daniel-MacDougall, Ph.D., associate professor of Epidemiology and senior author of the study.
After skin cancer, prostate cancer is the most common cancer in men in the United States. Since most cases are low-risk disease, localized to the prostate and have favorable outcomes, many men do not need immediate treatment and opt for active surveillance by their doctor. Treatments for prostate cancer can cause changes in quality of life and declines in urinary and sexual function, therefore there is interest in finding modifiable factors for men managed by active surveillance.
The study followed 410 men on an active surveillance protocol with Gleason grade group 1 or 2 localized prostate cancer. All study participants underwent a confirmatory biopsy at the beginning of the study and were evaluated every six months through clinical exam and laboratory studies of serum antigen PSA and testosterone.
Trial participants were 82.9% Caucasian, 8.1% Black and 9% other or unknown. The median age was 64, 15% of the men were diabetic and 44% used statins.
The men completed a 170-item baseline food frequency questionnaire, and Mediterranean diet score was calculated for each participant across 9 energy-adjusted food groups. The participants were then divided into three groups of high, medium and low adherence to the diet.
After adjustments for age and clinical characteristics, researchers saw a significant association between high baseline diet score and lower risk of cancer grade progression. For every one-point increase in the Mediterranean diet score, researchers observed a >10% lower risk of progression. After a median follow-up of 36 months, 76 men saw their cancer progress.
The study was limited by the low number of events in these men with mostly low risk disease monitored at MD Anderson. Future research is needed to see if the same effects are seen for larger and more diverse patient groups and men with higher-risk prostate cancer.
“Our findings suggest that consistently following a diet rich in plant foods, fish and a healthy balance of monounsaturated fats may be beneficial for men diagnosed with early-stage prostate cancer,” Gregg said. “We are hopeful that these results, paired with additional research and future validation, will encourage patients to adapt a healthy lifestyle.”
This research was supported by the Department of Defense Prostate Cancer Research Program Early Career Award (W81XWH-18-1-0193), a National Cancer Institute Cancer Center Support Grant to MD Anderson (CCSG 5P30 CA016672-37) and a Research Training Award for Cancer Prevention Post-Graduate Training Program in Integrative Epidemiology from the Cancer Prevention & Research Institute of Texas (RP160097). A full list of co-authors and disclosures can be found with the full paper here.
Newswise — Wildfire smoke contains microbes, a fact that’s often ignored, but one that may have important health repercussions.
In a perspective essay published in Science, Leda Kobziar and George Thompson call the attention of the scientific community to the health impacts of wildfire smoke’s microbial content.
Smoky skies caused by wildland fires are becoming seasonal norms, especially in some parts of the United States and Australia. In 2020, raging wildfires in the Western U.S. have set new records and led to extremely unhealthy or hazardous air quality levels for many weeks in a row.
It’s well-documented that exposure to wildfire smoke can damage the heart and lungs. Respiratory allergic and inflammatory diseases, including asthma and bronchitis, are also worsened by smoke exposure.
“The health impact of inhaling wildfire smoke increases dramatically during high-emissions wildfires and with long exposure,” said Kobziar, associate professor of Wildland Fire Science at the University of Idaho. “Yet, the risk of infection to the respiratory tract after this exposure is frequently overlooked.”
What role do microbes in wildfire smoke play in the spread of disease?
Wildland fire is a source for bioaerosol, airborne particles made of fungal and bacterial cells and their metabolic byproducts. Once suspended in the air, particles smaller than 5 μm can travel hundreds or even thousands of miles. Their movement depends on the fire behavior and the atmospheric conditions. Eventually, they are deposited or inhaled.
Bacteria and fungi can be transported in these wildland fire smoke emissions. While microbial concentration in smoke is higher near the fire source, these microbes may be active agents spreading infection. For example, coccidioidomycoses - a fungus that becomes airborne when soils are disturbed- is the cause of Valley fever, a potentially serious infection.
“We don’t know how far and which microbes are carried in smoke,” said Thompson, associate professor of Clinical Medicine at UC Davis. “Some microbes in the soil appear to be tolerant of, and even thrive under, high temperatures following wildfires.”
As Kobziar explained, “At the scale of a microbe, fire behavior research has shown that heat flux is highly variable, so it may be that many microbes aren’t even subjected to the high temperatures for very long. They may also be protected in small clusters of particulate matter.”
Kobziar and Thompson proposed a multidisciplinary approach to understanding the nature of the relationship between microbes, wildfire smoke and health. The complexity of the phenomenon calls for the expertise of scientists from different fields such as fire ecology, environmental microbiology, epidemiology, atmospheric sciences and public health and infectious disease.
“With longer wildfire seasons and higher severity trends, there is an urgency to work together in studying the behavior of the microbes carried by the smoke and their impact on human health,” Thompson said.
Article: Kobziar & Thompson, (2020). Wildfire smoke: A potential infectious agent, Science, DOI: 0.1126/science.abe8116
Newswise — Temperature data collected by wearable devices worn on the finger can be reliably used to detect the onset of fevers, a leading symptom of both COVID-19 and the flu, according to a team of researchers from the University of California San Diego, UC San Francisco and MIT Lincoln Lab.
Researchers published their results in a paper titled “Feasibility of continuous fever monitoring using wearable devices” in the Dec. 14 issue of the journal Scientific Reports. They emphasize that the study is a proof-of-concept effort with data from only 50 participants reporting COVID-19.
The Scientific Reports paper is the first published result from TemPredict, a study of more than 65,000 people wearing a ring manufactured by Finnish startup Oura, that records temperature, heart rate, respiratory rate and levels of activity. The goal of the study is to develop an algorithm that can predict the onset of symptoms such as fever, cough and fatigue, which are characteristic of COVID-19. Researchers say they hope to reach that goal by the end of the year. They also hope the algorithms will allow public health officials to act faster to contain the virus’ spread.
“This isn’t just a science problem, it’s a social problem,” said Benjamin Smarr, the paper’s corresponding author and a professor in the Department of Bioengineering and the Halicioglu Data Sciences Institute at UC San Diego. “With wearable devices that can measure temperature, we can begin to envision a public COVID early alert system.”
But users from diverse backgrounds would need to feel safe sharing their data for such efforts to really work, Smarr added. The data is stripped of all personal information, including location, and each subject is known by a random identifying number.
Smarr is TemPredict’s data analytics lead. Ashley Mason, a professor in the Department of Psychiatry and the Osher Center for Integrative Medicine at UC San Francisco, is the principal investigator of the study.
“If wearables allow us to detect COVID-19 early, people can begin physical isolation practices and obtain testing so as to reduce the spread of the virus,” Mason said. In this way, an ounce of prevention may be worth even more than a pound of cure.”
Wearables such as the Oura ring can collect temperature data continuously throughout the day and night, allowing researchers to measure people’s true temperature baselines and identify fever peaks more accurately. “Temperature varies not only from person to person but also for the same person at different times of the day,” Smarr said.
The study, he explains, highlights the importance of collecting data continuously over long periods of time. Incidentally, the lack of continuous data is also why temperature spot checks are not effective for detecting COVID-19. These spot checks are the equivalent of catching a syllable per minute in a conversation, rather than whole sentences, Smarr said.
In the Scientific Reports paper, Smarr and colleagues noticed that fever onset often happened before subjects were reporting symptoms, and even to those who never reported other symptoms. “It supports the hypothesis that some fever-like events may go unreported or unnoticed without being truly asymptomatic,” the researchers write. “Wearables therefore may contribute to identifying rates of asymptomatic [illness] as opposed to unreported illness, [which is] of special importance in the COVID-19 pandemic.”
The 50 subjects in the study all owned Oura rings and had had COVID-19 before joining TemPredict. They provided symptom summaries for their illnesses and gave researchers access to the data their Oura rings had collected during the period when they were sick. The signal for fever onset was not subtle, Smarr said. “The chart tracking people who had a fever looked like it was on fire.”
The data collected as part of the subsequent TemPredict study included 65,000 subjects, and these data will be stored at the San Diego Supercomputer Center at UC San Diego, where a team led by Ilkay Altintas is building a portal to enable other researchers to access these data for other analyses.
“The data collected has great potential to be linked with other datasets making individual and societal scale models be combined to further understand the disease,” said Ilkay Altintas, the chief data science officer at the San Diego Supercomputer Center, who is . The easier we can make to share the data and optimize the use of it through digital technologies, the quicker other researchers will make use of it in their studies.”
Researchers also are keeping up efforts to recruit a diverse pool of subjects that reflects the U.S. population.
“We need to make sure that our algorithms work for everyone,” Smarr said.
In future, researchers plan to expand their early detection methods to other infectious diseases, such as the flu.
Smarr has worked as a consultant with Oura within the last 12 month and received compensation, although not during this research project.
Feasibility of continuous fever monitoring using wearable devices
Benjamin Smarr, UC San Diego Department of Bioengineering and Halicioglu Data Science Institute
Kirstin Aschbacher, UC San Francisco and Oura
Sarah M. Fisher, Anoushka Chowdhary, Kerena Puldon, Adam Rao, Frederick Hecht and Ashley E. Mason UCSF
Stephan Dilchert, City University of New York and preValio LLC, Minneapolis
Photo Credit:
Oura
While not an FDA registered healthcare device, the Oura ring monitors a range of signals, including continuous temperature, heart rate, respiration rate and activity. Initial analysis suggests that a destabilization of temperature happens a couple of days before coronavirus symptoms manifest. The Oura ring detects this pattern.
Newswise — DETROIT – Henry Ford Health System is the first in the country to perform a procedure using the CG-100™ intraluminal device, which is temporarily inserted into the gastrointestinal tract and designed to reduce diverting stoma rates, and the need for an ostomy bag, in patients undergoing gastrointestinal resection procedures due to colorectal cancer treatment.When a patient has a cancerous part of their intestine surgically removed, the intestinal tract must be reconnected. While the surgically reconnected intestinal tract is healing, patients typically require a stoma, also known as an ostomy, which is a surgical opening on their abdomen that connects to their digestive tract. The stoma diverts digestive waste into an ostomy bag outside the abdomen and keeps it away from the site of reconnection. The ostomy must be reversed when it is no longer needed, which requires additional surgery and recovery time for the patient.Created by Colospan Ltd., the CG-100 device is a silicone sheath that is introduced into the intestinal tract through the rectum, no surgery required, and covers the site where the intestinal tract has been reconnected. This sheath aims to prevent or reduce the contact of fecal material with the site of reconnection, avoiding the need for a stoma. After 10 days, when the risk for leakage is reduced, the sheath is removed without any additional surgery.“One of the most serious complications that can happen after part of the gastrointestinal tract is surgically removed is a leak at the site of the resection,” said Craig Reickert, M.D., division head of Colon and Rectal Surgery at Henry Ford Cancer Institute. “While an ostomy procedure can reduce the risk of this type of leak, it is invasive and can be challenging for the patient to live with. One of the greatest potential benefits of this device is that it not only reduces the need for a stoma, it also does not require additional surgery to implant or remove.”The first CG-100 procedure, which was performed at Henry Ford Hospital by colon and rectal surgeon Surya Nalamati, M.D., is part of a Food and Drug Administration Investigational Device Exemption clinical trial that is comparing the CG-100 device to a diverting stoma, the current standard of care treatment for colorectal surgery. Henry Ford is the only site in Michigan, and one of just 12 sites nationwide, currently enrolling colorectal cancer patients in this clinical trial.The CG-100 clinical trial is multi-center and randomized, so patients who meet study requirements and agree to enter the study are randomized to either be treated with the CG-100 intraluminal bypass device or to receive a diverting stoma. The patient’s care team will continue to follow up with them for up to 39 weeks after surgery.According to the Centers for Disease Control and Prevention, in 2017 – the latest year for which incidence data are available – 141,425 new cases of colorectal cancer were reported in the United States, and 52,547 people in the U.S. died of this cancer. While an ostomy procedure can be lifesaving for colorectal cancer patients, it also carries with it risks of complications, such as incisional hernia, surgical site infection and anastomotic stenosis.To learn more about colorectal cancer treatment at Henry Ford Cancer Institute or to request an appointment at with a colorectal cancer specialist, visit henryford.com/services/colon-rectal-cancer.
Photo Credit: Colospan Ltd.
The CG-100™ intraluminal bypass device is temporarily inserted into the gastrointestinal tract and designed to reduce diverting stoma rates, and the need for an ostomy bag, in patients undergoing gastrointestinal resection procedures due to colorectal cancer treatment.