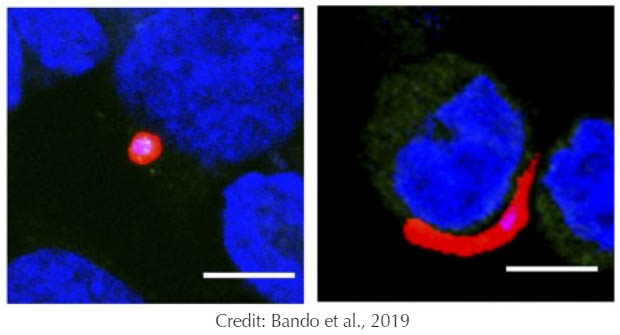
In normal human liver cells (left), Plasmodium parasites (red) develop into a circular, exoerythrocytic form that gives rise to malaria. But in cells lacking CXCR4 (right), the parasite remains trapped in its rod-shaped sporozoite form.
Newswise — Researchers in Japan have discovered that the Plasmodium parasites responsible for malaria rely on a human liver cell protein for their development into a form capable of infecting red blood cells and causing disease. The study, which will be published June 12 in the Journal of Experimental Medicine, suggests that targeting this human protein, known as CXCR4, could be a way to block the parasite’s life cycle and prevent the development of malaria.
According to the World Health Organization, there were an estimated 219 million cases of malaria in 2017, resulting in the deaths of approximately 435,000 people. Infected mosquitoes transmit Plasmodium parasites to humans in the form of rod-shaped sporozoites that travel to the liver and invade liver cells (hepatocytes). Once inside these cells, the Plasmodium sporozoites develop into spherical exoerythrocytic forms (EEFs) that eventually give rise to thousands of merozoites capable of spreading into red blood cells and causing malaria.
“It seems likely that the transformation of Plasmodium sporozoites into EEFs is tightly controlled so that it only occurs in hepatocytes and not at earlier stages of the parasite’s life cycle,” says Masahiro Yamamoto, a professor at the Research Institute for Microbial Diseases of Osaka University. “However, we know very little about the host factors that regulate the differentiation of sporozoites in infected hepatocytes.”
In the new study, Yamamoto and colleagues discovered that a hepatocyte protein called CXCR4 helps Plasmodium sporozoites transform into EEFs. Depleting this protein from human liver cells reduced the ability of sporozoites to develop into EEFs. Moreover, mice pretreated with a drug that inhibits CXCR4 were resistant to malaria, showing reduced levels of parasites in the blood and significantly higher survival rates following Plasmodium infection.
Yamamoto and colleagues also identified a cell signaling pathway that causes hepatocytes to produce more CXCR4 in response to Plasmodium infection and determined that the protein aids the parasite’s development by raising the levels of calcium inside the cells.
“Our study reveals that CXCR4 blockade inhibits Plasmodium sporozoite transformation in hepatocytes,” Yamamoto says. “Most anti-malaria drugs targeting Plasmodium-derived molecules eventually lead to drug resistance in these parasites. However, inhibitors targeting human proteins such as CXCR4 might avoid this problem and could be used prophylactically to prevent the development of malaria. Moreover, the CXCR4 inhibitor used in this study is already widely used in humans undergoing treatment for blood cancers, which could accelerate its repurposing as a new way of combating malaria.”
Bando et al. 2019. J. Exp. Med. http://jem.rupress.org/cgi/doi/10.1084/jem.20182227?PR

